To continue as a company that contributes broadly to people’s health, we are constantly pursuing the challenge of creating high-value new drugs that meet medical needs.
Therefore, in tandem with our in-house drug discovery innovation, we are expanding the development pipeline through in-licensing from external parties.
We are also working to stabilize existing business alliances and create new businesses through out-licensing to secure earnings, with the aim of supporting the continuous growth of Kyorin’s new drugs business.
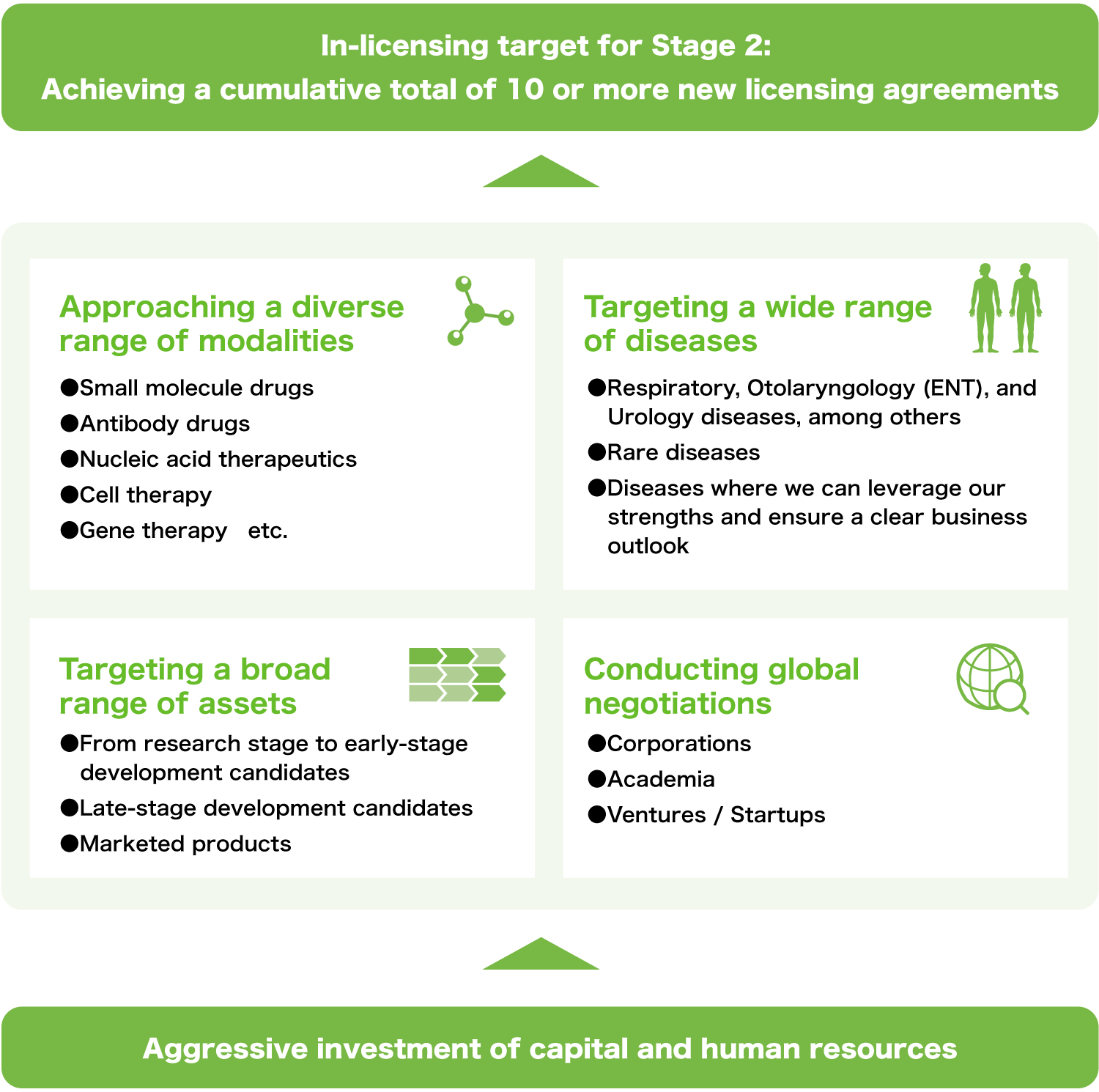
During the period of the Medium-Term Management Plan "Vision 110 —Stage 1—," we secured seven in-licensed products. Furthermore, under the Medium-Term Management Plan "Vision 110 —Stage 2—," we aim to achieve a cumulative total of 10 or more in-licensed items, thereby further solidifying our business foundation.
Expanding our development pipeline through proactive in-licensing investment
Try for drug innovation through new drug discovery strategies
- Engaging in a wide range of licensing activities, regardless of modalities or therapeutic areas
- Utilizing all available means to ensure the successful acquisition of in-licensed products
Promoting global out-licensing activities
We are proactively pursuing out-licensing activities with global companies to maximize the value of our proprietary products. In October 2020, we concluded an agreement to transfer the intellectual property rights of the immunomodulator KRP-203 to Priothera of Ireland; in March 2021, we signed a licensing agreement with Eisai to develop and sell the overactive bladder treatment Vibegron (sales name in Japan: Beova) in four ASEAN countries; and in March 2023, we signed a licensing agreement with Sumitomo Pharma to develop, manufacture, and sell Vibegron in Taiwan and other regions.
We also concluded a global licensing agreement with Novartis of Switzerland in March 2025 for our proprietary KRP-M223. Going forward, we will continue to engage in proactive partnering activities worldwide to quickly roll out our own products in various countries and regions to provide high-value pharmaceutical products that contribute to people’s health.
Partnering with Companies in Japan and Overseas



