The environment for drug discovery is becoming increasingly complex in terms of technologies related to new drug development due to changes including diversification in modalities and basic technologies and the spread of digital transformation (DX).
While these drug discovery technologies are becoming more sophisticated and achieving them becoming increasingly difficult, Kyorin aims to contribute to people’s health by pursuing new drug discovery strategies and drug discovery innovation for the continuous creation of high-value new products that meet medical needs.
One specific success was the out-licensing of our proprietary product KRP-M223 to Novartis in March 2025. We strongly hope that global development will move forward and that this product will help many patients around the world. Going forward, in addition to our core technologies in small molecule drug discovery, we will proactively use new external technologies to create new drugs that provide new value.
We will also actively work with external organizations with a focus on human resources development by raising researchers’ level of specialization and broadening their perspectives, with the aim of achieving the long-term vision “Vision 110.”
Changing environment
- Increasing sophistication, difficulty, and higher costs of drug discovery
- Diversification and complexity of drug discovery modalities and basic technologies
- Evolution and proliferation of digital technologies
Opportunities
- Development of basic research technologies to increase drug discovery research opportunities
- Acceleration of research through activation of open innovation
- Use of big data and AI to streamline R&D
- Increase in new treatment options thanks to digital technologies
Risks
- Increasingly competitive environment due to development of research technologies and acceleration of environmental changes
- Rise in development costs due to stricter clinical trials and stricter approval of new drugs
- Market contraction due to reform of drug pricing system and its impact on business viability
Medium-term business plan
Vision 110 –Stage1– initiatives
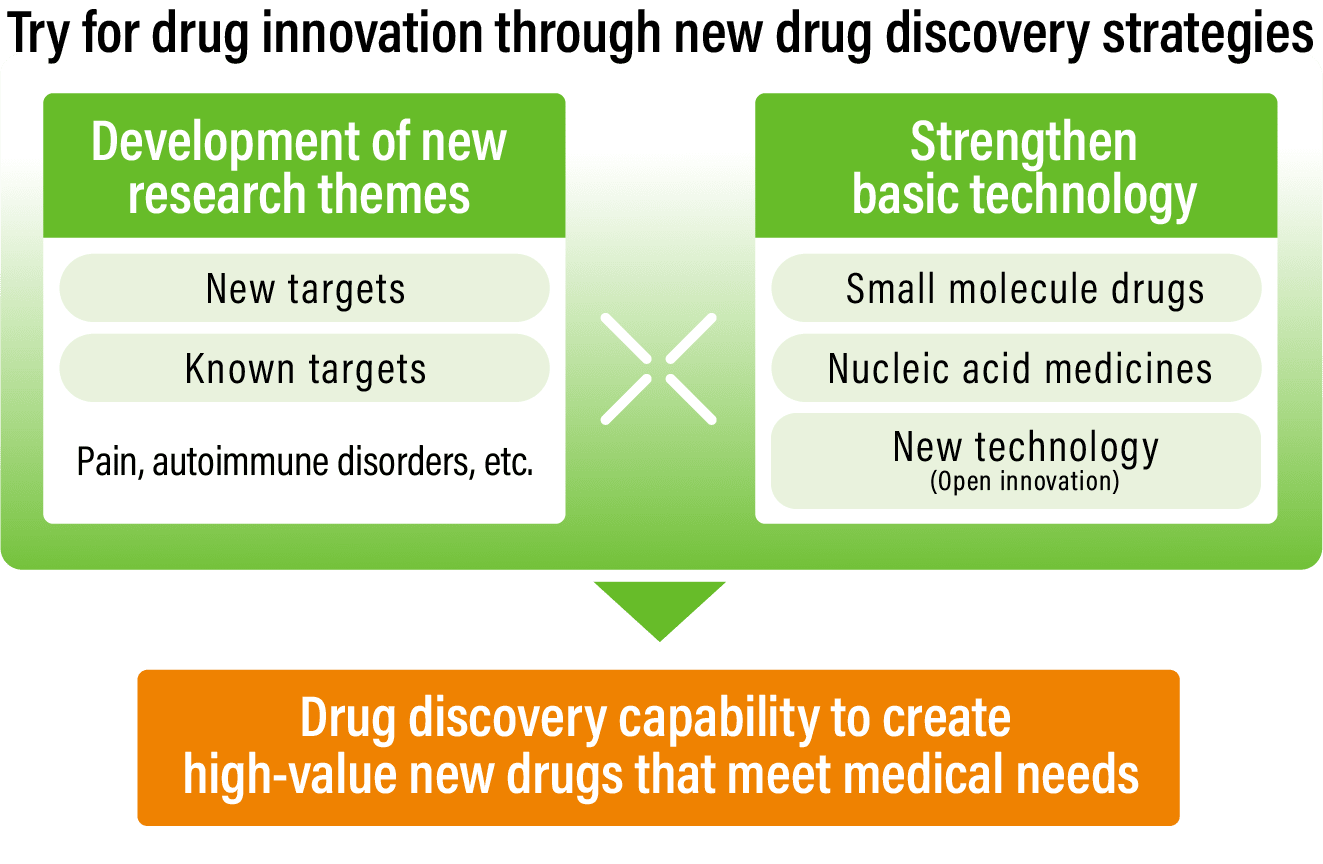
Business strategyStrengthening drug discovery capability to create high-value new drugs that meet medical needs
Pursue drug innovation through new drug discovery strategies
- Engage in drug discovery using novel technologies for existing treatments that have issues, in addition to drug discovery for diseases where drug contribution is low
- Combine drug discovery technologies and disease research to create new high value drugs
- Drug discovery technologies: Work to deploy nucleic acid drug discovery and external technologies in addition to small molecule drug discovery
New drug discovery strategies
In the Group’s core business of new drugs, the continuous creation of new drugs has become an important issue. To resolve this, we recognize that identifying drug discovery programs and formulating research and development strategies targeting future products are essential. We have strengthened our organizational functions to achieve these goals.
By combining drug discovery technologies and disease research (drug discovery targets), we are taking on the challenge of “drug discovery innovation” that creates new value.
We are engaged in research and development to create value in response to unmet medical needs by working to create new drugs to address diseases where drug contribution is low and also by carrying out drug discovery that demonstrates clinical significance by deploying new technologies to address issues with existing treatments.
To make maximum use of our drug discovery capabilities, we are concentrating management resources in specific areas of drug discovery research (pain, autoimmune disorders, etc.) to identify and pursue drug discovery programs more effectively.
The out-licensing of our proprietary product KRP-M223 to Novartis in March 2025 is one example of our drug discovery capabilities bearing fruit. We expect this achievement to lead to global development going forward.
Strengthening collaboration with external organizations and pursuing research themes
In terms of drug discovery technologies, besides building on our core technologies in small molecule drug discovery, we are examining nucleic acid drug discovery and proactively incorporating external technologies. In January 2024, we concluded a joint research agreement with Veneno Technologies Co. Ltd., under which we are carrying out a program to obtain functional disulfide-rich peptides (DRPs) using Veneno’s next-generation peptide discovery technology. By looking beyond our own technologies and ideas and proactively incorporating superior external research and technologies, we aim to create new drugs that offer new value.
We will engage in selection and concentration of research themes that create value while formulating and verifying exit strategies. In the initial exploratory research stage, we will pursue drug discovery activities that emphasize scientific approaches to create and achieve target therapeutic profiles. After optimization research into leading compounds, we will use target product profiles to decide whether to move forward.
Expanding development pipeline through in-licensing and formulating development and medical strategies to maximize value
We will further strengthen cooperation between relevant departments to accelerate the evaluation and acquisition of in-licensing candidates, while formulating development strategies from a unique perspective with a constant awareness of novel clinical evaluation methods and therapeutic strategies for development candidates.
We envision diversifying our modalities and pursuing global development as we expand our development pipeline, including for in-licensed products. This approach will allow us to develop unique strategies while strengthening our regulatory function.